How Depression Can Be Treated with a Psychedelic Trip
If you’re interested in psychedelics, then you might have heard of the work of Robin Carhart-Harris, who conducted much of the most relevant research in the world of psychedelics together with his team at Imperial College in London.
In this look back at ICPR 2016 we will highlight the talk he held about his team’s trials with psychedelics-assisted psychotherapy, where he also showed some beautiful visuals of his team’s brain research, which happened to become some of the most famous psychedelic brain imagery known on the internet.
ㅤ
ㅤ
Like our upcoming ICPR 2022 near Amsterdam, the edition in 2016 strove to bring together as many relevant studies from psychedelics as possible, and Carhart-Harris’ talk was most certainly a highlight. His research has been cited often and his talk was one of the best-watched from that year’s ICPR on our Youtube channel.
In his talk, Carhart-Harris talks about the results of his research – that psychedelics can cause a rise in cognitive flexibility, neuroplasticity, creative thinking, imaginative suggestibility, emotional lability, positive moods, and optimism.
He also touches on the idea of depressive realism, a trend he has seen in patients suffering from depression. He describes their depression as a “sort of delusion”, where his patients “don’t see the world as it really is. There is this really quite evident pessimism bias, that is normalised post-treatment with psilocybin.”
ㅤ

ㅤ
A testimony of one of the participants is featured in the talk:
ㅤ
26:35 — ‘Although it’s early days yet, the results are amazing. I feel more confident and calm than I have in such a long time. My outlook has changed significantly too, I’m more aware that it’s pointless to get wrapped up in endless negativity. I also feel as if I’ve seen a much clearer picture. [Now] I can enjoy things the way I used to, without the cynicism, without the oppression. At its most basic. I feel like I used to before the depression.”
ㅤ
Brain Scans
One way to go about investigating psychedelics is by making fMRI brain scans. These scans are made of healthy and depressed individuals before, during and after a psychedelic experience. This way, the brain can be observed for changes.
Through these scans, the team got insights into the inner workings of the brain during psychedelic trips, and how they correlate with described experiences of volunteers, like ego-death. This is a type of experience in which people who are under the influence of psychedelics describe a certain loss of self, and a deeper connection with the wider universe or nature.
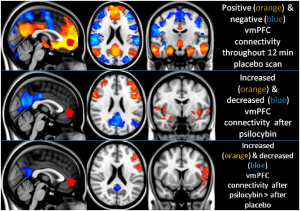
Carhart’s studies have highlighted that the Default Brain Network may be connected with our sense of self – our ego – and that the lower activity of this network during a psychedelic session may be associated with the occurrence of ego-death.
ㅤ
ㅤ
12:40 — “We see quite reliably a relationship between the magnitude of the disintegration and the default brain network. [..] The greater the disintegration of the default mode network, the greater our volunteers’ ratings of ego-dissolution. ”
ㅤ
During the psychedelic experience induced with psilocybin, the parts of the brain associated with the Default Brain Network show a drastic reduction in activity, often creating the experience of ego-death. The compulsive activity of the Default Brain Network also has been associated with patients that scored higher in depression ratings.

The Default Brain Network and the Self
Robin Carhart-Harris’s argument is that the Default Brain Network may be the source of what most adult people call the ‘ego’. This network is known as the Default Mode Network because, during our daily lives, this brain network becomes more active when we are idle.
The Default Mode is actually a really important part of our mental stability. This network is responsible for keeping our routines in check, making sure that our pending matters stay afloat, and that we’re not overlooking anything.
The mental activity generated by the Default Mode Network is usually stable and consistent day after day. This daily consistency in addition to the fact the DMN is the ‘standard’ mental voice, may contribute to the illusion that the Default Mode Network is the self.
ㅤ
12:58 — [The Default Brain Network is]: “Arguably the best candidate we have for the neural substrates of the self, or the ego, or our identity and personality.” – Robert Carhart-Harris
ㅤ
By analyzing the brains of participants who consumed psilocybin, Carhart’s team noticed that there was a process of renewal happening within the structure of the brain, almost like a general mind reset. This process of rebirth has been reported many times by psychedelic subjects.
ㅤ
17:50 — “We can think of the mind or the brain is reset in the same way that you can think of a computer is malfunctioning and throwing up an error message and you are wondering what you can do. And then you press the reset button and it comes back working nice and smooth as it should.”
ㅤ
In more recent years, Carhart-Harris has worked on building a more unified model of the workings of psychedelics in the brain. He founded the Psychedelic Research Group at Imperial College in London and focuses on the action of psychedelic drugs in the brain, and their clinical utility as aides to psychotherapy, with a particular focus on depression. He still studies the brain effects of LSD, psilocybin (magic mushrooms), and MDMA.
Robin Carhart-Harris will not be speaking at ICPR 2022, but his colleague and the new head of the Psychedelic Research Group at Imperial College will: David Nutt.
Notes about the author: Alexandre Perrella is a writer for Cabbanis!
How Depression Can Be Treated with a Psychedelic Trip Read More »

